Water treatment refers to any technique that enhances the quality of water to be used for a specified purpose. The end-use could be drinking, industrial water supply, irrigation, river flow maintenance, water recreation, or any other applications, including safely returning to the environment. Water treatment removes or reduces the concentration of pollutants and undesired components so that the water is fit for its intended end-use. This therapy is critical for human health since it allows people to benefit from both drinking and irrigation.
Drinking water treatment
Treatment for drinking water production entails removing pollutants and/or inactivating any potentially hazardous organisms from raw water to produce pure enough water for human use without any risk of ill health effects in the short or long term. The largest microbiological dangers are connected with drinking water polluted with human or animal feces (including bird feces). Pathogenic bacteria, viruses, protozoa, and helminths can all be found in feces.
The removal or elimination of microbial pathogens is critical. This typically requires reactive chemical agents such as suspended solids to eliminate bacteria, algae, viruses, fungi, and minerals such as iron and manganese. These toxins continue to wreak havoc on several developing countries that lack access to safe drinking water.
Water quality measures include the treatment of the water and its transportation and distribution following treatment. As a result, it is usual to practice leaving residual disinfectants in treated water to fight bacterial contamination during distribution and maintain the pipes clean.
Water delivered to residential properties for use as tap water or other purposes may be further treated before consumption, often with an in-line treatment procedure. Water softening and ion exchange are two examples of such treatments. Many proprietary systems claim to be able to remove leftover disinfectants and heavy metal ions as well.
Also Read, 9 Lesser-Known Benefits Of Drinking Warm Water
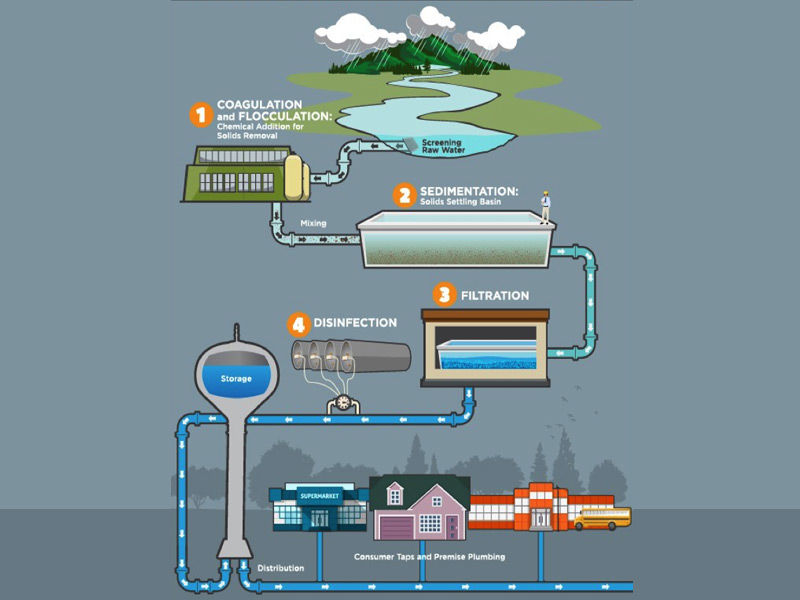
Procedure
- Physical procedures like settling and filtration, chemical processes such as disinfection and coagulation, and biological processes such as slow sand filtration are used to remove contaminants.
- For municipal drinking water treatment worldwide, a mixture of the following methods (depending on the season and pollutants and chemicals present in the raw water) is utilized.
Chemicals
- Pre-chlorination is used to suppress algae and stop biological growth.
- When there is a minor amount of manganese present, aeration and pre-chlorination are used to remove the dissolved iron.
- Decontamination with chlorine, ozone, and ultraviolet light to kill bacteria, viruses, and other pathogens.
Physical
- The removal of dissolved particles trapped in the floc by sedimentation for solids separation.
- Filtration removes particles from water by passing it through a sand bed that can be washed and reused or by passing it through a specially designed filter that may be washable.
- To remove suspended particles, dissolved air flotation is used.
Biological
- A biofilm is used in slow sand filtration to decompose organic debris, absorb soluble components, and entrap particles.
- Coagulation in preparation for flocculation.
- Coagulant aids, also known as polyelectrolytes, are used to improve coagulation and floc formation.
- Polyelectrolytes, often known as polymers in the field, often have either a positive or negative charge; what is employed is entirely dependent on the source water properties of the treatment facility.
- These are frequently employed in conjunction with a Primary Coagulant like ferric chloride, ferric sulfate, or alum.
Conventional Treatment

Technological advances
Technologies for potable water and other purposes are well developed, and generalized designs from which treatment methods can be selected for pilot testing on the specific source water are available. Several commercial companies also offer unique technology solutions for the treatment of specific pollutants. Water treatment automation is ubiquitous in the developed countries. Source water quality, scale, and environmental effects can all influence capital and operating expenses. The end use of the treated water defines the necessary quality monitoring systems. The level of automation utilized is often dictated by locally available expertise.
Desalination process
Freshwater can be produced by treating saline water. Reverse osmosis and distillation are the two basic procedures employed. Both procedures demand more energy than local surface water treatment. They are typically employed exclusively in coastal locations or water, such as groundwater, with high salt.
Portable Water Purification
Living distant from drinking water sources frequently necessitates the use of a portable water treatment technique. These can range in complexity from the simple insertion of a disinfection tablet to a hiker’s water bottle to complex multi-stage processes delivered to disaster zones by boat or plane.
Also read, Seven Unique Yet Breathtaking Waterfalls That End Up Into The Sea
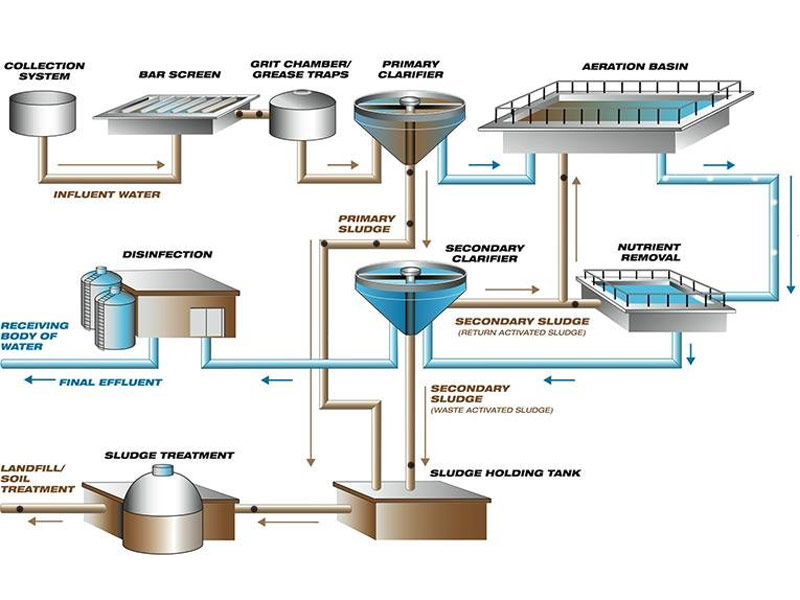
Industrial Water Treatment process
Procedures
Boiler water treatment and cooling water treatment are two of the most important industrial water treatment processes. A significant amount of adequate water treatment can result in the reaction of particles and bacteria within the pipework and boiler housing. When left untreated, steam boilers can develop scale or corrosion. Scale deposits can generate weak and unsafe machinery. At the same time, increased thermal resistance necessitates more fuel to heat the same amount of water. Poor quality filthy water can provide a breeding ground for germs such as Legionella, putting public health at risk.
Dissolved oxygen, acidity, and excessive alkalinity can all induce corrosion in low-pressure boilers. As a result, water treatment should eliminate dissolved oxygen while maintaining the boiler water’s pH and alkalinity values. A cooling water system that is not properly treated might suffer from scale formation, corrosion, and fouling, as well as becoming a breeding ground for hazardous microorganisms. This decreases efficiency, shortens the life of the plant, and makes operations unreliable and dangerous.
Boiler water treatment
Boiler water treatment is a sort of industrial water treatment that focuses on removing or chemically modifying contaminants that may be harmful to the boiler. To eliminate scaling, corrosion, or foaming, different methods of treatment are applied in different areas. External treatment of raw water sources intended for use in a boiler focuses on impurity removal before the water reaches the boiler. Internal treatment within the boiler focuses on decreasing the ability of water to dissolve the boiler and keeping contaminants in the least likely forms to create problems before they can be removed from the boiler during boiler blowdown.
Cooling water treatment
Water cooling is a technique for removing heat from components and industrial equipment. Where air cooling is ineffective, water may be a more efficient heat transfer fluid. In most populated areas, water provides the thermal conductivity benefits of a liquid with an abnormally high specific heat capacity and the possibility of evaporative cooling. Because of the low cost, single-use coolant loops are frequently rejected as waste, however, recycling coolant loops can be pressured to prevent evaporative loss and offer more portability and enhanced cleanliness.
Technologies
Chemical therapy
Chemical treatments are methods used to make industrial water fit for use or discharge. These include chemical precipitation, chemical disinfection, chemical oxidation, advanced oxidation, ion exchange, and chemical neutralization.
Physical therapy
Filtration eliminates particles from water by passing it through a layer of sand, such as in a fast gravity filter, or bypassing it through a mechanical filter.
Dissolved air flotation is a method of removing suspended materials from water. This is achieved by dissolving air under force in water and then discharging the water/air mixture at atmospheric pressure in a flotation tank. The released air creates microscopic bubbles that stick to the suspended particles, causing them to rise to the water’s surface to be removed by a skimming device or an overflow.
Biological therapy
Slow sand filters purify raw water using a biological process to generate potable water. They function by utilizing a complicated biological layer that naturally forms on the surface of the sand. The hypogeal layer, also known as the Schmutzdecke, is a gelatinous biofilm found in the upper few millimeters of the sand layer. The surface biofilm cleans the water as it runs through the layer. At the same time, the underlying sand acts as a support medium for the biological treatment layer.
Bacteria, fungus, protozoa, Rotifera, and various aquatic insect larvae make up the Schmutzdecke. More algae may form as the biofilm ages, and larger aquatic animals such as bryozoa, snails, and Annelid worms may be present. Water percolates through the hypogeal layer, trapping matter particles in the mucilaginous matrix and adsorbing soluble organic material. Bacteria are microorganisms. Fungi and protozoa metabolize the pollutants.

























